What is the "premature oxidation" issue exactly?
Several consumers, producers and critics have reported that a non-negligible number of bottles of white burgundies suffer from a phenomenon called “premature oxidation,” often referred to as “premox.” Simply put, the wines appear at various stages of oxidation, and this state is not what would normally be expected given their age. Although there are technically two chemically distinct types of premature oxidation (which are discussed below), the typical signs of oxidation include a darker color than expected as well as distinct “oxidation” aromas or flavors (sometimes to the point of making the wine completely undrinkable).
Oxidation occurs in stages. In the “partial oxidation” stage, the wine usually has a color which is somewhat darker or more mature gold than the wine’s vintage counterparts and either a notable absence of the wine’s normal aromas and flavors and a “flat “ character on the palate, or the wine also exhibits aromas and/or flavors associated with oxidation – typically brown apple, cereal grains (often described as “cheerios” or “corn flakes”), toasted bread (as distinct from oak toast), light sherry-like notes or sometimes aromas like well-aged cognac. Aromas and flavors which we frequently associate pleasantly with fully mature wines, such as honey, butterscotch, nutty or hazelnut, are also associated with oxidation of wine. When these particular characteristics are more prominent than expected in a relatively young wine, along with advanced color, this too is usually a sign of partial oxidation. Partially oxidized wines are often still drinkable (and occasionally very enjoyable) despite the noticeable oxidative flaws. In the more advanced stages of oxidation, the wine is usually a very full blown gold or orangey-gold color or sometimes quite brown and usually exhibits very prominent cereal grain, overly-toasted bread, or strong sherry-like aromas and flavors that make the wine virtually undrinkable.
Several theories have been put forward to explain this phenomenon but none of them is fully satisfying and convincing for the time being. However it seems clear that there is no one single cause and that the issue stems from a combination of aggravating factors (see below for a discussion of each suggested cause).
There are Two Distinct Types of Oxidation In Bottled Wine
The oxidation of bottled wine actually consists of two distinct but chemically coupled types of oxidation reactions. One type of oxidation results in the color changes we associate with premature oxidation. The other type of oxidation results in the changes in aromas and flavors referred to above.
The first type of wine oxidation is oxidation of flavonoid phenols, which are polyphenolic compounds located in the grape skins, seeds and stems. This type of phenolic compound is generally associated with pressing or extractive techniques. Thus, wines which are subject to more vigorous pressing exhibit considerably higher levels of flavonoid phenols and wines which are made with very gentle pressing and no extractive techniques exhibit very little in the way of flavonoid phenols. When flavonoid phenols are oxidized they result in color changes in the wine (browning), which change the wine over time from bright yellow to increasingly deeper shades of gold and finally brown color. There is another group of phenolic compounds in grape juice, called non-flavonoid phenols, which make up the largest amount of the phenols present in white wine and are found primarily in the pulpy part of the grape. While non-flavonoid phenols also oxidize over time in wine, they do not produce any detectable changes in color, aromas or flavors.
The second type of wine oxidation is oxidation of ethanol, the alcohol produced by the fermentation process. The ethanol is oxidized by hydrogen peroxide (H2O2) present in the wine, which results in acetaldehyde. The primary source of hydrogen peroxide in the wine is from the oxidation of phenols. When acetaldehyde reaches the threshold detection level (generally between 8 and 10 ppm depending on the individual), we perceive the oxidized aromas and/or flavors described above.
While the combination of an oxidized color and oxidized aromas and flavors most frequently occur simultaneously, it is certainly possible to have a wine with a brilliant youthful-appearing color which exhibits oxidized aromas and flavors. Conversely, it is also possible to have a wine which exhibits considerable browning but no oxidized flavors or aromas. Personally, I’ve experienced more than one example of each. Moreover, it is also possible to reduce or eliminate the color changes which normally take place over time by means of a controversial technique known as “hyper-oxidation,” or sometimes more gently referred to as “browning of the must,” which is discussed below.
There is a chemical interrelationship between the two types of oxidation. Wine chemists refer to this as “coupled oxidation.” One interrelationship between the two different oxidation processes is that the oxidation of phenols in the wine creates hydrogen peroxide (H2O2) as a byproduct which, unless it reacts with free sulfur dioxide (free SO2), will oxidize ethanol into acetaldehyde. Thus, the oxidation of phenols eventually leads to the oxidation of ethanol once the free SO2 is exhausted. A second interrelationship is that the phenols prevent the oxidation of ethanol that would otherwise directly occur from free sulfate radicals derived from the SO2 added to the wine. Thus, from the perspective of oxidation of ethanol into acetaldehyde, there is a mutual interdependence between the phenols and the SO2 added to the wine to prevent oxidation. The third interrelationship relates to the phenols ability to absorb or remove oxygen as the phenols are oxidized. Due to some unique chemical properties, phenols in the wine act both as substrates for oxidation (in layman’s terms, as vehicles for oxidation or as the component which is actually oxidized) and as buffers to the oxidation of ethanol (in layman’s terms, as inhibitors to the oxidation of ethanol.) It is generally agreed that the flavonoid and non-flavonoid phenols in the wine are the primary buffer or barrier to the oxidation of ethanol in white wine. Sulfur dioxide is also a barrier to oxidation, but its role is to react with and effectively remove hydrogen peroxide.
The incidence of premature oxidation by vintage
The premature oxidation problem, which began with the 1995 vintage, was not really recognized as a systemic problem until the fall of 2003, when the 1996 vintage reached seven years of age. At that time a small but discernable percentage of the wines began to exhibit premature oxidation symptoms similar to those which the 1995 vintage began exhibiting about a year earlier. When an inordinately high number of the 1995 vintage whites appeared to be partially or fully oxidized, it seemed inexplicable and people naturally sought to explain the problem as somehow related to the vintage (and there had been a considerable amount of rot in 1995). When the 1996 vintage started to show the same problems, despite being considered an ultra-clean, high acid and classic vintage, alarm bells started going off. Each subsequent vintage has exhibited similar problems beginning about 6.5 to 7 years after the vintage (although the vintages subsequent to 1999 have a lower incidence of outright oxidation than do 1995, 1996 and 1999, the incidence of advanced wines has been higher in a majority of the subsequent vintages.)
The first public commentary about the premature oxidation problem occurred in September of 2004 in Issue 116 of Steven Tanzer's International Wine Cellar, in conjunction with Mr. Tanzer's annual white burgundy review. Two months later, in Issue 16 of Burghound, Allen Meadows had an extended discussion of the issue. Thereafter other wine critics began to publicly acknowledge that there was in fact a problem. The initial reaction of most burgundy producers was complete denial that their wines were affected.
Now thirteen years after the problem was first publicized, premature oxidation has clearly reached epidemic proportions. The problem clearly affects every white burgundy vintage between 1995 and 2009 (i.e. each vintage which has reached seven and a half years of age).
For example, the results of the annual vintage tastings/oxidation checks I have conducted each of the last nine years show the following variation in incidence of premature oxidation:
1996 vintage (tasted at 10 years of age):
Corked: 2/28 (7%)
Oxidized: 5/28 (18%)
Advanced: 3/28 (11%)
Total advanced plus oxidized: 8/28 (29%)
1999 vintage:
Corked: 1 or 2 of 44 (2% to 4%)
Oxidized: 9/44 (20%) Highest percentage of oxidized wines ever
Advanced: 3/44(7%)
Total Oxidized + Advanced: 12/44 (27%)
2000 vintage (tasted at 7 and 7.5 years of age):
Corked: 1/58 (2%)
Oxidized: 9/58 (16%)
Advanced: 7/58 (12%)
Total Oxidized + Advanced: 16/58 (28%)
2001 vintage:
Corked: 0/43 (0%)
Oxidized: 4/43 (9%)
Advanced: 7/43 (16%)
Total Oxidized + Advanced: 11/43 (26%)
2002 vintage:
Corked: 3/64 (5%)
Oxidized: 5/64 (8%) or 6/64 (by my count) (9%)
Advanced: 5/64 (8%) Tied for lowest percentage of advanced wines ever
Total oxidized + advanced: 11/64 (17%)
2004 vintage:
Corked: 1/63 (2%)
Permanently Reduced: 1/63 (2%)
Oxidized: 3/63 (5%) Lowest percentage of oxidized wines ever
Advanced: 5/63 (8%) Tied for lowest percentage of advanced wines ever
Total Oxidized + advanced: 8/63 (13%) Lowest total percentage ever
2005 vintage:
Corked: 1/66 (2%) [Corked bottle of Raveneau MDT replaced on night one]
Oxidized: 4/65 (6%) Second lowest percentage of oxidized wines
Advanced:16/65 (25%) Highest percentage of advanced wines ever
Total Oxidized + advanced: 20/65 (31%) Highest total percentage ever
2006 vintage:
Corked: 0 or 1 of 28: 0% to 4% (includes 3 California ringers)
Oxidized: 0/25 white burgundies (initial group consensus) or 2/25 (my late run back through all wines) 0% to 8%
Advanced: 3/25 white burgundies (12%)
Total Oxidized +advanced: 5/25 (20%)
2007 vintage:
Corked: 1/71 (1.4%)
Oxidized: 3/71 (4.22%)
Advanced - 6/71 (8.45%) or by my count 9/71 (12.68%)
Total Oxidized + advanced: 9/71 12.68% (lowest group total ever) or 12/71 (my count) 16.9%
2008 vintage:
Corked: 1/70 (1.4%)
Other consensus defects: 2/70 (2.9%)
Oxidized: 3/70 (4.29%)
Advanced - 9/70 (12.9%) or by my count 10/70 (14.3%)
Total Oxidized + advanced: 12/70 (17.1%) or by my count 13/70 (18.6%)
2009 vintage:
Corked: 0/62 (0%)
Otherwise Chemically “Off” 1/62 (1.6%)
Oxidized: 2/62 (3.2%)
Advanced: 6/62 (9.7%)
Total Oxidized + advanced: 8/62 (12.9%)
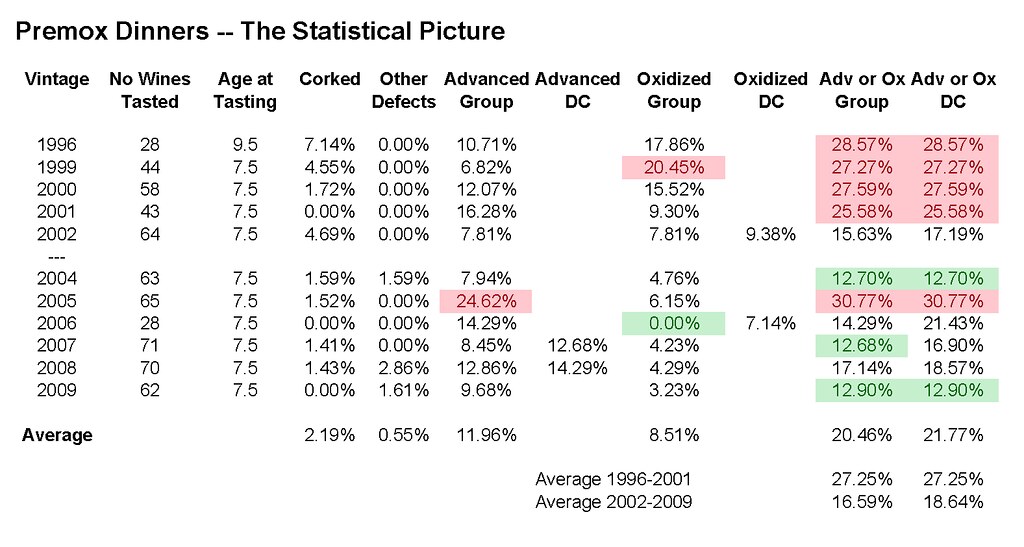
The most interesting pattern to emerge here is that the vintages from 2002 and thereafter have a lower average combined percentage of oxidized and advanced wines than the vintages from 1996 through 2001.. But the one really notable exception is the 2005 vintage which had the worst performance ever.
While the problem is clearly an epidemic one, it has received scant attention in the mainstream press. There has been no serious coverage of this issue in the Wine Spectator's print issue and the issue was never discussed in the print version of the Wine Advocate until December of 2009. One can only speculate why. There have been articles in the London Times and Le Figaro (Paris) mentioning this wiki site, as well as mentions in a number of other newspapers and articles. This issue has slowly come to public consciousness in Europe and while most American wine collectors are quite familiar with the issue, it surprisingly remains unknown to many casual wine drinkers in the US .
Some producers are clearly more affected than others
The oxidation which has been observed in the 1995 to 2009 vintages seems to vary widely among producers. Some producers seem to have a very high incidence of oxidized bottles while others have very little. See the following link which discusses the varying incidence of premox among the producers. Premox Incidence Among Producers
Oxidation will most often vary within a single case
The oxidation problem also varies among single cases of wine. Sometimes several bottles within the same case are severely affected whereas the other bottles in the same case are showing perfectly well (or at least according to expectations). While the cause or causes of premature are still subject to debate, it would appear that the highly variable performance of bottles within in a single case is explained by an unexpectedly high variation in the oxygen permeablity of corks. Empirical data from different studies has demonstrated a large variance in the oxygen permeability of randomly selected high quality wine corks of the same grade. (For additional details, see the discussion of lowered sulphur dioxide levels as a potential cause.)
Philippe Prost, the winemaker at Bouchard, has stated that he has observed a considerable variation in weights among corks included in even the highest grades of cork used on Bouchard's best wines. As he points out, the corks with lower weights necessarily mean that the corks in question are less dense and more porous. Prost believes, properly I think, that the corks with lower weights have the greatest potential to allow too much oxygen into the bottle and thus premature oxidation. To overcome this problem Bouchard has recently begun weighing each cork (for those corks to be used on Bouchard's premium bottlings) and rejecting any corks found to have substandard weight. In essence, Bouchard is attempting to “slice off” the worst portion of the bell curve on oxygen permeability of corks. For this Bouchard is to be highly commended. Hopefully, this is a trend that will quickly catch on.
Distinguishing oxidation which isn't "premature"
All white wines, even when stored in ideal storage, will eventually oxidize. In some cases, properly cellared bottles of Montrachet from great vintages have been known to survive 100 years or more without being completely oxidized. While grand cru wines generally have longer life expectancies than premier cru wines, which in turn generally have longer life expectancies than village wines, the vintage conditions have a significant impact on the expected life of a given vintage of white burgundy. Generally speaking, vintages which have high acidity levels (and lower ph) will survive much longer than average and, conversely, vintages with lower than average acidity levels will be naturally shorter-lived. Similarly, vintages impacted by rot, which produces enzymes in the wine that can accelerate the oxidation of ethanol (what we perceive as oxidation) can be relatively short-lived.
While there seems to be an accepted standard among burgundy collectors that a typical well-made grand cru white burgundy from a decent vintage should last an absolute minimum of 10 years without any signs of oxidation (and longer in the case of high acid vintages like 1993 and 1996), it's simply not reasonable to apply this same type of expectation for vintages recognized as poor or seriously rot-affected vintages (such as 1997 and 1998). Thus, I'm troubled when I hear someone complaining that their eight to ten year old 1997 or 1998 burgundy was “prematurely” oxidized. My view is that while the wine was oxidized, it wasn't “premature” for the wine to fail at eight to ten years of age given the lousy quality of the vintage to start with; or, stated alternatively, it simply wasn't reasonable to expect the wine to be exhibiting no oxidation when it was eight-plus years old. For similar reasons, I'm troubled when someone occasionally claims that a 1989 or 1990 white burgundy that the person recently tasted was “prematurely” oxidized.
To me at least, it's simply not a useful excercise to talk about “premature” oxidation of vintages like 1997, 1998 or 2003 because the expected life span of the vintage was quite short. Similarly, I think it's pointless to talk about “premature” oxidation of wines older than 15 years.
The potential causes of premature oxidation
Several reasons have been outlined to explain the situation. However each of them has a number of counter-examples, which points to a combination of factors as the most likely cause.
Below are listed a number of potential factors contributing to the premature oxidation of white burgundies, each of which is discussed in some detail.
- Batonnage (stirring of the lees)
Footnotes:
[Text and opinions by Editor Don Cornwell. © Don Cornwell 2005-2017]